Posted by [email protected]

PDCA stands for Plan Do Check Act. The PDCA cycle is a four-step quality management method used in business for the control and continuous improvement of processes and products. It’s also known as the Deming cycle, the Shewhart cycle, the control cycle, or PDSA (Plan Do Study Act).
This cycle is helpful for beginning a new project such as introducing a new product, new equipment, new production line, and so one. It is also helpful for defining a repetitive work process, such as on an assembly line. Additionally, you can use the PDCA cycle when making any kind of change in your process – whether as a result of a corrective action or as part of your facility’s continuous improvement process. The PDCA Cycle is also the bedrock of implementing or streamlining food safety schemes, such as FSSC 22000. Let’s now take a closer look!
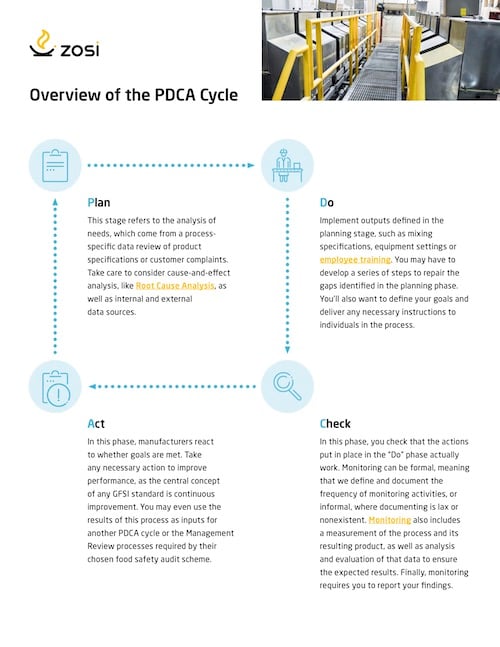
Overview of the PDCA Cycle
Plan
This stage refers to the analysis of needs, which come from a process-specific data review of product specifications or customer complaints. Take care to consider cause-and-effect analysis, like Root Cause Analysis, as well as internal and external data sources.
Do
Implement outputs defined in the planning stage, such as mixing specifications, equipment settings or employee training. You may have to develop a series of steps to repair the gaps identified in the planning phase. You’ll also want to define your goals and deliver any necessary instructions to individuals in the process.
Check
In this phase, you check that the actions put in place in the “Do” phase actually work. Monitoring can be formal, meaning that we define and document the frequency of monitoring activities, or informal, where documenting is lax or nonexistent. Monitoring also includes a measurement of the process and its resulting product, as well as analysis and evaluation of that data to ensure the expected results. Finally, monitoring requires you to report your findings.
Act
In this phase, manufacturers react to whether goals are met. Take any necessary action to improve performance, as the central concept of any GFSI standard is continuous improvement. You may even use the results of this process as inputs for another PDCA cycle or the Management Review processes required by their chosen food safety audit scheme.
Conclusion
Whatever step you are taking toward continuous improvement in your facility, the PDCA cycle and procedures act as an essential tool for starting. Learn more about the PDCA cycle’s use in GFSI schemes – like FSSC 22000 – here.
Download our new print-and-go PDCA cycle infographic below.


 Never Miss an Update.
Never Miss an Update. 